Ionizing radiation, such as the flux of electrons, alpha particles, neutrons, gamma rays, etc., is used in radiochemistry. Ionizing radiation loses energy when absorbed by the substance.
The rad is used to measure absorbed energy doses. A rad is worth 100erg/g, which means that one gram of substance has absorbed 100erg of energy. The ionizing capacity of X or gamma rays is measured in roentgens. A roentgen corresponds to a radiation intensity that produces 2 08x10-9 ion pairs in a cubic cm of dry air at 0°C and under 760mm of Hg (corresponding to a mass of 0.001293g of air).
A roentgen is equal to 87.7 ergs of energy that one gram of air can absorb. The radioactive isotopes used as radiation sources include radon, polonium, and radium, which emit alpha and gamma rays naturally, as well as synthetic isotopes like tritium, cesium 137, cobalt 60, phosphorus 32, strontium 90, sulfur 35, and others that emit beta rays.

The AGLAE ion accelerator at the Research Center in France.
- It takes special equipment (particle accelerators like betatrons, cyclotrons, etc.) to produce strong fluxes of electrons or cations.

70 MeV Cyclotron installed at the University of Nantes (France)
Nuclear reactions can be used to generate neutron fluxes, for instance:
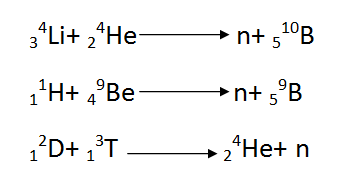
[Created by using word microsoft]
Detection and measurement of radioactivity:
The Geiger counter, which is used to measure a lot of ionizing radiation, is a very useful measuring device. A brief current pulse between the wire and the tube walls is produced by the ions and electrons that are created when a particle ionizes the gas in the Geiger counter tube. Thus, each particle that enters the tube is recorded as the pulse is amplified and connected to a recorder at its output.
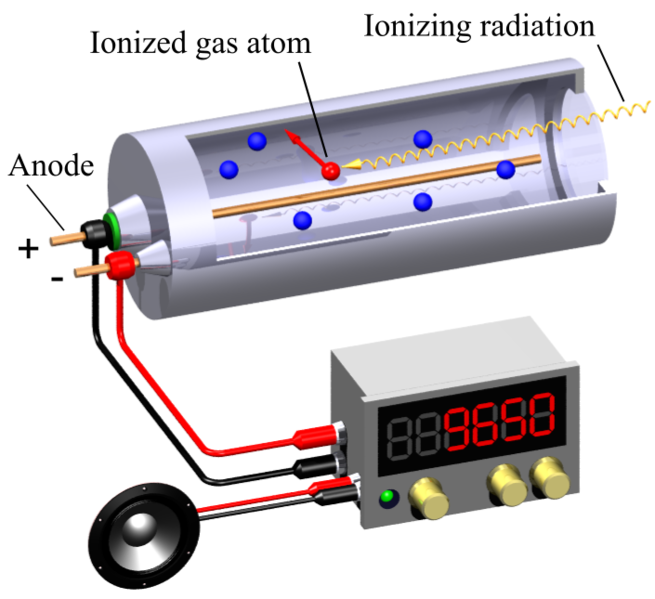
Geiger Muller counter. A detector of radiation levels
Nuclear particles can be detected using other ingenious methods such as fluorescent screens and photographic film. Scintillations are caused when nuclear particles strike screens coated with fluorescent materials such as zinc sulfide and barium platinocyanide.
Still essential are photography-based detection techniques. Photographic film is worn by scientists when they are near radioactive sources; the amount of radiation exposure is revealed by the blackening of these films. In some specialized emulsions, the alpha particle's path can be fixed, highlighting the significant advancement in photographic emulsion quality.
References:
[Smail Meziane: Livre Chimie générale- Structure de la matiére. Berti edition, Alger, 2006]
[Principles & Applications of Photochemistry, Brian Wardle,Wiley, ISBN 0470014938]
[R. OUAHES et B. DEVALLEZ- Chimie générale- Office des publacations universitaires- Alger]
T. Mill- Reactions and Processes: Chemical and Photo Oxidation
Detection and Measurement of Radioactivity- David J. Malcolme-Lawes BSc, PhD, CChem, FRIC
Thanks for your contribution to the STEMsocial community. Feel free to join us on discord to get to know the rest of us!
Please consider delegating to the @stemsocial account (85% of the curation rewards are returned).
You may also include @stemsocial as a beneficiary of the rewards of this post to get a stronger support.
Congratulations @benainouna! You have completed the following achievement on the Hive blockchain and have been rewarded with new badge(s):
Your next target is to reach 15000 upvotes.
You can view your badges on your board and compare yourself to others in the Ranking
If you no longer want to receive notifications, reply to this comment with the word
STOPCheck out the last post from @hivebuzz:
Support the HiveBuzz project. Vote for our proposal!
Congratulations @benainouna! You received a personal badge!
You can view your badges on your board and compare yourself to others in the Ranking
Support the HiveBuzz project. Vote for our proposal!