In one of my previous posts, I had presented one of the most advanced ongoing trials concerning the SARS-CoV-2 vaccine.
We have seen that the research is conducted by the Jenner Institute of Oxford University with the partnership of the Italian company ADVENT-IRBM of Pomezia.
And we have underlined how the use of adenovirus seems to be, after the first tests, a solution capable of effectively promoting the production of antibodies without causing related damages.
In this post, we will see in more detail what is the mechanism of action that researchers are proposing, so far, with good feedback in ChAdOx1 nCoV-19, the Italian-British vaccine candidate.
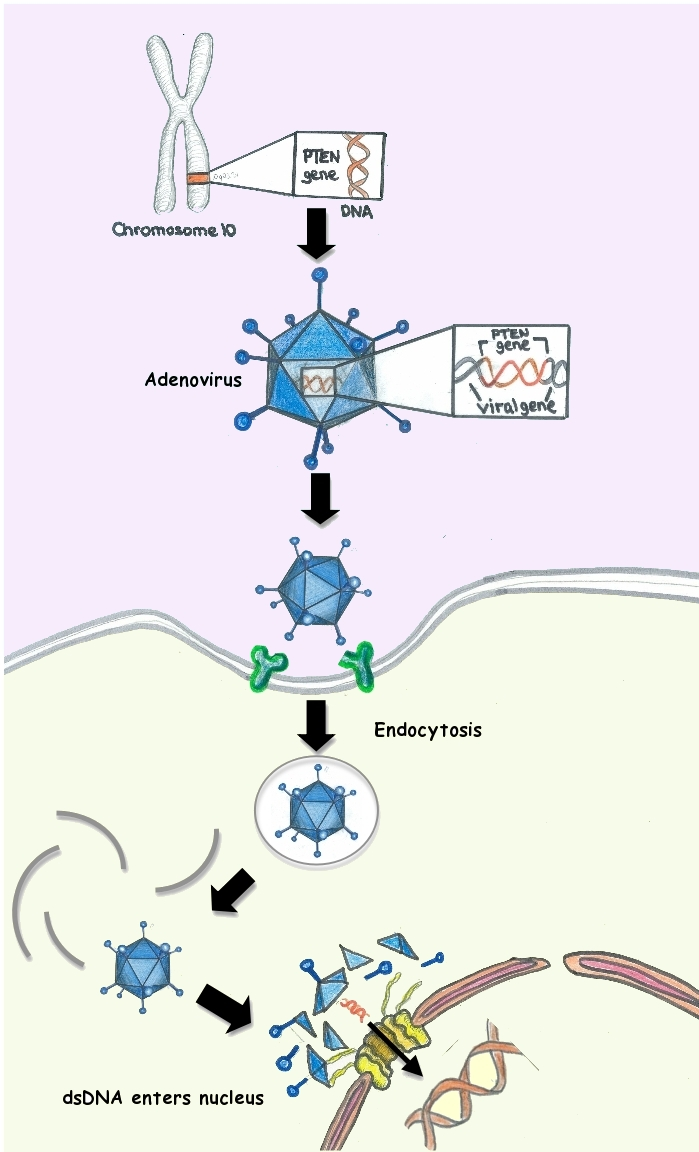
Image CC BY-SA 3.0
Using a virus such as Trojan horse to carry a gene capable of triggering the immunization process against SARS-CoV-2 within the body, is a strategy that closely resembles that used in gene therapy.
And in fact this can be called a genetic vaccine.
In general, the vaccine is a preventive tool against various pathologies which, in its most classic form, consists of inactivated or attenuated viruses or bacteria, capable of eliciting a reaction from the immune system.
In the first case, these are dead organisms (which therefore cannot cause disease), in the second, organisms rendered harmless (similarly incapable of causing disease) but modified to provoke a response which generally consists in the production of antibodies.
A genetic vaccine, on the other hand, is capable of inducing both an antibody and cellular response.
In practice, to defeat pathogens, the immune system has to produce both antibodies to it and cells, especially CD4 and CD8 lymphocytes to attack it.
Basically, if some virus manages to escape the antibodies and enter the cells, this other weapon of the immune system can intervene.
The classic example of a genetic vaccine can be represented by a simple DNA fragment that codes for a surface protein of the pathogen. The most significant obstacle is being able to get it inside the cell because, as is known, DNA is unlikely to cross the cell membrane.
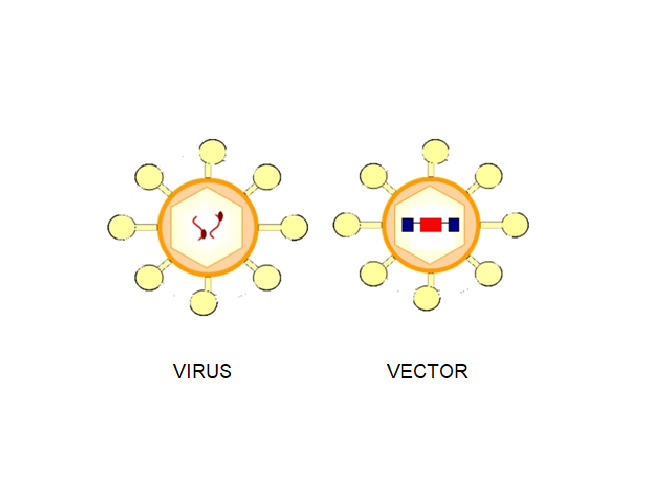
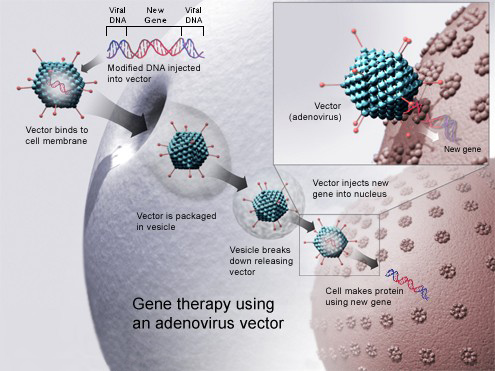
A virus can represent an excellent vector for DNA, since inside the viral vector a piece of DNA can be inserted which corresponds to the protein of the pathogen against which the individual is to be vaccinated.
Thus, through its specific receptors, the viral vaccine enters and can activate the whole immune system, generating an effective antibody and cellular response against the aggressor.
Adenoviruses have a genome consisting of a double strand of DNA of about 35 kb, 30 of which can be replaced with the gene of interest.
Once inside the cell, adenovirus does not integrate into the genome but replicates itself in the nucleus like an episode.
Despite having small DNA, they can carry large inserts (36Kb), they are also easy to manipulate in the laboratory and safe, because, as mentioned, they do not integrate into the human genome.
Another advantage is that they are not neutralized by antibodies because we all have them more or less, as adenoviruses are very common in the population. Fact that makes them even more powerful.
For all the reasons listed, they are perfect platforms for vaccine development.
Public Domain
ChAdOx1 nCoV-19 exploits an adenovirus derived from a chimpanzee made harmless by depriving it of the gene responsible for replication (E1), in the place of which the gene corresponding to the spike protein of the SARS-CoV-2 virus is inserted. In this way, the adenovirus is no longer able to replicate.
At this point, to produce it in large quantities, they resort to cell lines in which the E1 gene is present and complements the missing gene.
At this production stage, only an increase in intact virus molecules containing the DNA piece of the spike protein is required. This becomes possible by exploiting the replication apparatus of the cell line.
The system must be forced to produce only the virus molecules, in order to have a sufficiently high number in the next phase. At the end of the growth phase, in which the volume of the cell mass has grown, the adenoviruses that carry the piece of DNA of the spike protein must be separated from the other cellular components and this is accomplished by collecting the cells and lysing them in the only solution that will be made go through a chromatographic column capable of retaining the adenoviruses and instead let everything else pass.
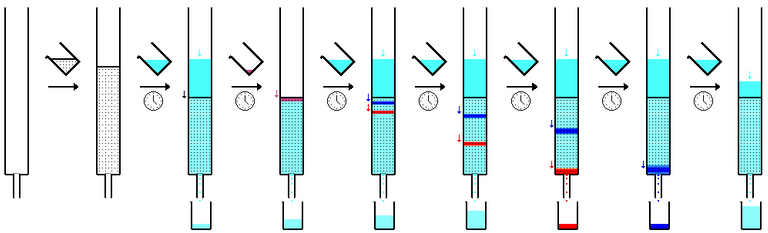
Column chromatography sequence - Public Domain
This is the purification phase, at the end of which the adenovirus is obtained which will be further subjected to sterilization and filtration to free it from any further bacteria and microorganisms.
The final preparation is a liquid and sterile solution, which can be administered by intramuscular injection.
Adenoviruses represent a platform already demonstrated in adults and children without adverse reactions.
However, the timing of release of the vaccine will not be very short.
Although the vector has already been tested on animals and humans, in this case it contains a new piece of DNA inside it, and in reality it represents a new molecule that needs to be re-tested for safety and immunogenicity.
Various tests have been carried out in the laboratory to ensure that the preparation is free of contaminants or other viruses, and still others will be conducted.
Only once its sterility has been ascertained is access to the release phase of the clinical batch for human studies entered.
In phase 1 of the clinical trial, the vaccine was administered to 510 healthy volunteers and the results are very positive indeed.
Phase 2 now begins, in which the vaccine will be tested on 5000 subjects.
The program also includes a phase 3 clinical trial with 30000 participants and, if everything goes smoothly, a large-scale deployment can begin in December 2020.

Thanks to all of you for reading my article.
If you liked it, share it on your blog and write me a comment with your impressions.
See you soon!
GM
Sources
- https://www.scienzainrete.it/articolo/luglio-si-prova-sulluomo-vaccino-italiano/cristiana-pulcinelli/2020-04-17
- https://www.insalutenews.it/in-salute/azienda-italiana-lavora-al-vaccino-contro-il-coronavirus-da-qui-allestate-produrra-1-000-dosi/
- https://www.osservatorioterapieavanzate.it/news/covid-19-italia-in-prima-linea-nello-sviluppo-di-un-vaccino-genetico
- https://www.news-medical.net/life-sciences/How-is-Adeno-Associated-Virus-Used-as-a-Viral-Vector
- https://it.wikipedia.org/wiki/Terapia_genica#Gli_adenovirus

Credits @doze
Does it mean that we are sure we get can immunity? This was still unclear, at least according to what I have read O(10) days ago.
We're still not sure, but we may be on track.
Clearly this is not the only vaccine being tested (I read that there are approximately 100 vaccine candidates in progress) and in the end we may have more than one.
You said (and read) well...in the last 10 days, phase 1 has been completed with the results on the administrations to the 510 volunteers.
So these are really recent news. In fact, it was not easy to find sources on the method of action of the vaccine, there is still no published study and not even a preprint.
The first results are positive, in September we will see how the experimentation on 5000 people went and then we will move on to phase 3.
Surely there will be priority for the funding countries and for the risk categories (doctors, nurses, elderly, immunosuppressed) but if the tests give good results, we could think of obtaining immunization in the short-medium term, i.e. by 2021.
Thanks for the clarifications! Hopefully, the virus won't mutate by then....
Yes, absolutely...with the vaccine we will be a little safer, but the battle will continue for a long time.
!discovery 40
Grazie per il supporto ✌️
I tuoi post sono sempre molto interessanti! :)
🙏🏼🤗
This post was shared and voted inside the discord by the curators team of discovery-it
Join our community! hive-193212
Discovery-it is also a Witness, vote for us here
Thanks for your contribution to the STEMsocial community. Feel free to join us on discord to get to know the rest of us!
Please consider supporting our funding proposal, approving our witness (@stem.witness) or delegating to the @stemsocial account (for some ROI).
Please consider using the STEMsocial app app and including @stemsocial as a beneficiary to get a stronger support.